Leann Tilley laboratory
-
Head of Laboratory, Redmond Barry Distinguished Professor of Biochemistry & Pharmacology, ARC Australian Laureate Fellow (2015-2020)

Professor Leann Tilley+61 3 8344 2227
Research Overview

Join us at Biomolecular Horizons 2024: Discover, Create, Innovate. To be held in Melbourne, Australia, from 22- 26 September, 2024
The Tilley lab is working as part of a global effort to understand and control malaria. We undertake research in the areas of cell biology and drug development related to the malaria parasite, Plasmodium falciparum.
Read our recent article, 'Reaction hijacking of tyrosine tRNA synthetase as a new whole-of-life-cycle antimalarial strategy' by Stanley C. Xie et al. 2022. Science chose this article as being of immediate relevance to public health concerns and has provided free access.
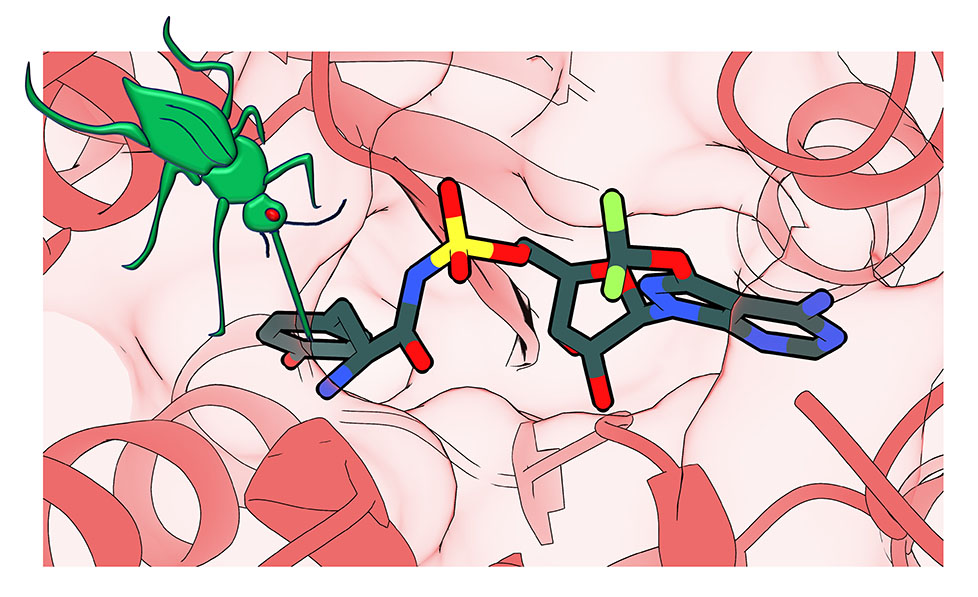
Diagrammatic representation of the target of a new antimalarial compound, ML901 (coloured structure), that shows highly specific and potent inhibition of the malaria parasite but is non-toxic to mammalian cells. [Image generated by Leann Tilley and Riley Metcalfe]
Malaria parasite-infected erythrocytes
Every minute, one child in Africa dies from malaria. Around the world, the mosquito-borne parasite kills about 450,000 people each year, most of them children and pregnant women, while another 200 million people suffer illness as a result of malaria infections.
Carried by the Anopheles mosquito, the tiny malaria parasite invades the victim's red blood cells where it starts devouring the red cell haemoglobin and changing the cells so that the infected red blood cells stick to blood vessel walls. This leads to the typical symptoms of fever and headaches, but in severe cases results in coma and death.
The Tilley lab is particularly interested in in designing antimalarial drugs, based on inhibition of protein translation. We are interested in how the parasite alters the erythrocyte surface to cause malaria pathology, as well as the remarkable transformation that turns parasites banana-shaped and allows them to be transmitted from a human host to a mosquito vector.
As part of her Australian Laureate Fellowship Program (2015-2020), with funding from the Australian Research Council, Professor Tilley created the Georgina Sweet Award for Women in Quantitative Bioscience. For further details:
Staff
Postdoctoral Fellows
- Stanley (Cheng) Xie
- Con Dogovski
Research Assistants
- Olivia Carmo
- Nutpakal Ketprasit
- Chia-Wei Tai
Graduate Students
- Jiahong Li
- Mohini Shibu
- Nutpakal Ketprasit
Research Publications
Click here for the results of a PubMed search of Leann's publications
Click here for the results of a Google Scholar analysis of Leann's publications
- Xie, S.C. Metcalfe, R.D., Dunn, E., Morton, C.J., Huang, S-C., Puhalovich, T., Du, Y., Wittlin, S., Nie, S., Luth, M.R., Ma, L., Kim, M-S., Pasaje, C.F.A., Kumpornsin, K., Giannangelo, C., Houghton, F.J., Churchyard, A., Famodimu, M.T., Barry, D.C., Gillett, D.L., Dey, S., Kosasih, C.C., Newman, W., Niles, J.C., Lee, M.C.S., Baum, J., Ottilie, S., Winzeler, E.A., Creek, D.J., Williamson, N., Parker, M.W., Brand, S., Langston, S., Dick, L.R., Griffin, M.D.W., Gould, A.E. and Tilley,L. (2022) Reaction hijacking of aminoacyl tRNA synthetases as a new anti-infectives strategy. Science 376, 1074–1079 (2022).
- Xie, S.C. Metcalfe, R.D., Mizutani, H., Puhalovich, T., Hanssen, E., Morton, C.J., Du, Y. Huang, S-C., Ciavarri, J., Hales, P., Griffin, R.J., Cohen, L.H., Chuang, B-C., Wittlin, S., Deni, I., Yeo, I., Barry, D.C., Liu, B., Gillett, D.L., Crespo-Fernandez, B.F., Ottilie, S., Mittal, N., Churchyard, A., Ferguson, D., Aguiar, A.C.C., Guido, R.V.C., Baum, J., Hanson, K.K., Winzeler, E.A., Gamo, F.J., Fidock, D.A., Baud, D., Parker, M.W., Brand, S., Dick, L.R., Griffin, M.D.W., Gould, A.E. and Tilley,L. (2021) Design of proteasome inhibitors with oral efficacy in vivo against Plasmodium falciparum and selectivity over the human proteasome. Proceedings of the National Academy of Sciences, USA 118 (39) e2107213118.
- Yang, T., Yeoh, L., Tutor, M., Dixon, M.W., McMillan, P.J., Xie, S.C., Bridgford, J.L., Gillett, D.L., Duffy, M., McConville, M.J., Ralph, S.A., Tilley, L., Cobbold, S.A., (2019) Decreased K13 abundance reduces haemoglobin catabolism and proteotoxic stress, underpinning artemisinin resistance. †Equal contribution Cell Reports 29, 2917-2928
- Xie, S.C.^, Metcalfe, R.^, Hanssen, E.^, Yang, T., Gillett, D.L., Leis, A.P., Morton, C.J., Kuiper, M., Parker, M.W., Spillman, N.J., Wong, W., Tsu, C., Dick, L.R., Griffin, M.D.W.* and Tilley, L.* (2019) The structure of the PA28/20S proteasome complex from Plasmodium falciparum and implications for proteostasis. Nature Microbiology 4(11):1990-2000. IF: 14.174. ^*Equal contribution.
- Bridgford, J.L., Xie, S.C., Cobbold, S.A., Pasaje, C.F.A., Herrmann, S., Yang, T., Gillett, D.L., Dick, L.R., Ralph, S.A., Dogovski, C., Spillman, N.J. and Tilley, L. (2018) Artemisinin kills malaria parasites by unfolding proteins and inhibiting the proteasome. Nature Communications 9, 3801. IF: 12.124
- Li, H., O'zDonoghue, A., van der Linden, W. A., Xie, S. C., Tilley, L., Craik, C. S., C. A. da Fonseca, P. and Bogyo, M. (2016) Structure and function based design of Plasmodium-selective proteasome inhibitors. Nature 530, 233-236.
- Dearnley, M.K., Trang, C.T.T., Zhang, Y., Looker, O., Huang, C., Klonis, N., Yeoman, J., Kenny, S., Arora, M., Osborne, J., Chandramohanadas, R., Zhang, S., Dixon, M.W.A. and Tilley, L. (2016) Host cell remodelling in sexual blood stages of the malaria parasite, Plasmodium falciparum. Proc. Natl. Acad. Sci. USA 113(17):4800-5.
- Dogovski, C., Xie, S.C., Burgio, G., Bridgford, J., Mok, S., McCaw, J., Chotivanich, K., Kenny, S., Gnädig, N., Straimer, J., Bozdech, Z., Fidock, D.A., Simpson, J.A., Dondorp, A.M., Foote, S., Klonis, N., Tilley, L. (2015) Targeting the cell stress response of Plasmodium falciparum to overcome artemisinin resistance. PLoS Biology 13(4): e1002132.
- Klonis, N., Xie, S.C., McCaw, J., Crespo, M., Zaloumis, S., Simpson, J., Tilley, L. (2013) Altered temporal response of malaria parasites determines differential sensitivity to artemisinin. Proc. Natl. Acad. Sci. USA 110:5157-62.
- Klonis N, Crespo M, Abu-Bakar N, Bottova I, Kenny S, Rosenthal P, Tilley, L (2011) Artemisinin action against Plasmodium falciparum requires hemoglobin uptake and digestion. Proc. Natl. Acad. Sci. USA 108:11405-10.
Research Projects
Faculty Research Themes
School Research Themes
Cellular Imaging & Structural Biology, Therapeutics & Translation, Infection & Immunity
Key Contact
For further information about this research, please contact Head of Laboratory, Redmond Barry Distinguished Professor of Biochemistry & Pharmacology, ARC Australian Laureate Fellow (2015-2020) Professor Leann Tilley
Department / Centre
Unit / Centre
MDHS Research library
Explore by researcher, school, project or topic.