Mike Griffin laboratory
-
Head of Laboratory

Dr Michael Griffin+61 3 8344 2534
Research Overview
Apolipoproteins and amyloid fibril formation in disease
Several diseases, including Alzheimer's, Parkinson's, diabetes type II and atherosclerosis are associated with the misfolding of proteins and the deposition of amyloid fibrils. Our work has focused on amyloid fibril formation by apolipoproteins (Fig. 1) in which we are developing strategies to understand and inhibit amyloid fibril formation. In particular we are interested in amyloid formation by apoA-I, which is associated with cardiovascular disease and hereditary amyloidosis. Our recent work has shown that oxidation of the methionine residues of apoA-I induces amyloid formation.
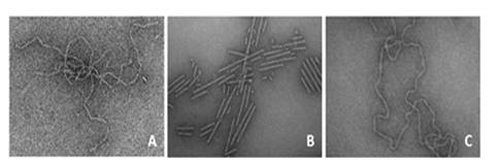
Figure 1: Transmission electron micrographys of amyloid fibrils formed by A. oxidized apoA-1, B. apoC-II in the presence of micellar phospholipid and C. lipid-free apoC-II
The structure of amyloid fibrils
Amyloid fibrils have increased β-structure relative to the non-fibrillar form and interact with the dyes Congo Red and thioflavin T (ThT). X-ray diffraction patterns indicate a common cross β-structure with strands in adjacent β-sheets oriented at right angles to the long axis of the fibril. Our NMR, X-ray diffraction and spectroscopic studies have been used to generate a simple 'G-like' structural model for apoC-II amyloid fibrils.
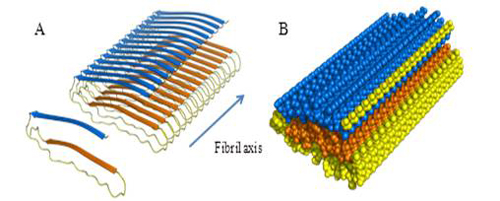
Figure 2: Structural model of apoC-II amyloid fibrils. A. ApoC-II monomer adopts a 'letter G-like' β-strand-loop-β-strand structure. Each monomer contributes to each of the two β sheets, giving rise to a parallel in-register structure. B. A space-filled view of apoC-II amyloid fibrils.
2 The structure and function of the Interleukin-11 signalling complex
Interleukin (IL)-11 is a cytokine protein implicated in the promotion of colon cancer. Little is known about the how IL-11 binds to cell surface receptors, or what genetic changes in these proteins predispose individuals to colon cancer.
Research Publications
Click here for the results of a PubMed search of Mike's publications.
Research Projects
For project inquiries, contact our research group head.
Faculty Research Themes
School Research Themes
Biomedical Neuroscience, Molecular Mechanisms of Disease
Key Contact
For further information about this research, please contact Head of Laboratory Dr Michael Griffin
Department / Centre
Unit / Centre
MDHS Research library
Explore by researcher, school, project or topic.