Lawson laboratory: Transmissible neurodegeneration
-
Head of Laboratory

Associate Professor Vicki Lawson+61 3 8344 4049
Research Overview
View Associate Professor Lawson's latest PubMed publications listing here.
Associate Professor Lawson recently spoke with ABC News about the long term effects of Covid-19. Read the article here.
The focus of the Lawson lab is understanding the pathogenesis of transmissible diseases affecting the central (CNS) and enteric nervous systems (ENS).
Under investigation are virological diseases such as COVID-19 and non-conventional infectious diseases including prion (Creutzfeldt Jakob disease) and prion-like diseases (synucleinopathies ie Parkinson’s disease). Of particular interest is the effect of strain variation on invasion, pathogenesis, and inflammation in the CNS and ENS. The aim of our research is to better understand disease pathogenesis to develop methods to treat, diagnose and prevent disease.
Creutzfeldt Jakob disease
Prion diseases are transmissible neurodegenerative diseases caused by the misfolding of the normal cellular prion protein. Diseases may be acquired, familial or arise sporadically and can affect parts of the brain important for memory, mood, and movement. We have developed mouse-adapted prion strains and use in vivo, in vitro, and cell-free models to understand how strain variation causes different forms of disease. From these models and in collaboration with the Australian Creutzfeldt Jakob disease registry we have identified novel features of human prion disease. The aim of our research is to better understand the pathogenesis of prion disease strains and develop methods to treat, diagnose and prevent disease.
Parkinson’s disease
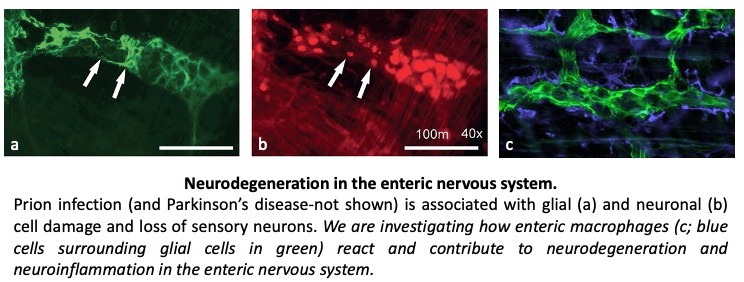
Parkinson's disease is a neurodegenerative disease associated with the misfolding and aggregation of alpha-synuclein. Recent studies have shown that the misfolding of alpha-synuclein can be propagated in a prion-like manner which has raised the possibility that Parkinson’s disease may also be a transmissible disorder. Parkinson’s disease is recognised as affecting motor function however early signs of disease also affect the function of the gastrointestinal tract. Our research has focussed on how Parkinson’s affects the enteric nervous system. We use in vivo, in vitro and cell-free models of familial and chemically induced Parkinson’s disease to understand how neurodegeneration contributes to the early signs of disease with the aim to develop early diagnostic and therapeutic methods to identify and prevent or slow the progression of disease to the central nervous system.
COVID- 19
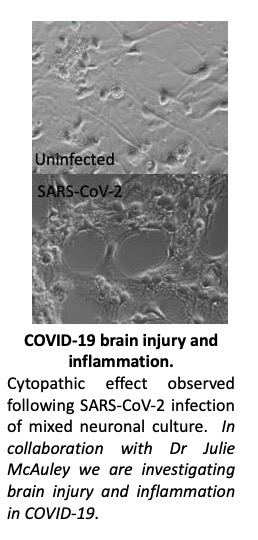
‘Brain fog’ is a term used to describe the lingering manifestations of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. Our research has focussed on the neuropathogenesis of SARS-CoV-2 Variants of Concern (alpha and delta variants) to understand the long-term clinical consequences of COVID-19. In collaboration with Dr Julie McAuley we have developed in vitro and in vivo infection models to identify the determinants of neuroinvasion, neuropathogenesis and neuroinflammation.
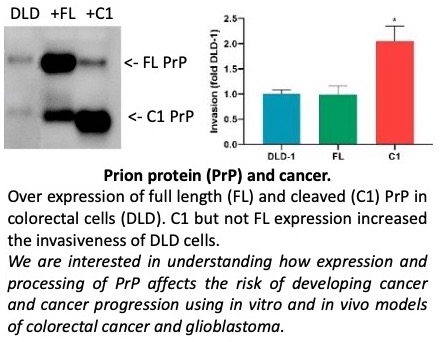
Prion protein and cancer
Misfolding of the normal cellular form of the prion protein leads to neurodegeneration, however over expression of the same protein has been linked to cancer. To understand the role of the prion protein in cancer we have investigated prion protein expression and processing using in vitro and in vivo models of colorectal cancer and glioblastoma. The results of our investigations have highlighted the importance of prion protein processing in cancer progression.
Staff
Connor Daymond, Research Project Student
Laura Ellett, PhD Student
Natalia Fortunado de Miranda, PhD Student
Yong Koo, MBiomed
Shana Portelli, PhD Student
Collaborators
Professor David Finkelstein, Florey Department of Neuroscience and Mental Health, The University of Melbourne
Professor John Furness, Department of Anatomy and Physiology, The University of Melbourne
Dr Julie McAuley, Department of Microbiology and Immunology, The University of Melbourne
Funding
2018-2021 NHMRC Project Grant: Unravelling the neuropathological basis of the gastrointestinal manifestations of Parkinson’s Disease (CIC).
The Lawson lab would like to acknowledge the CJDSGN for their ongoing support of our research.
Research Opportunities
This research project is available to PhD students, Masters by Research, Honours students, Master of Biomedical Science, Post Doctor Researchers to join as part of their thesis.
Please contact the Research Group Leader to discuss your options.
Research Publications
Cheng, L., Quek, C., Li, X., Bellingham, S. A., Ellett, L. J., Shambrook, M., Zafar, S., Zerr, I., Lawson, V. A., & Hill, A. F. (2021). Distribution of microRNA profiles in pre-clinical and clinical forms of murine and human prion disease. Commun Biol, 4(1), 411. https://doi.org/10.1038/s42003-021- 01868-x
Deacon, N. J., Tsykin, A., Solomon, A., Smith, K., Ludford-Menting, M., Hooker, D. J., McPhee, D. A., Greenway, A. L., Ellett, A., Chatfield, C., Lawson, V. A., Crowe, S., Maerz, A., Sonza, S., Learmont, J., Sullivan, J. S., Cunningham, A., Dwyer, D., Dowton, D., & Mills, J. (1995). Genomic structure of an attenuated quasi species of HIV-1 from a blood transfusion donor and recipients. Science, 270(5238), 988-991. https://www.ncbi.nlm.nih.gov/pubmed/7481804
Ellett, L. J., Hung, L. W., Munckton, R., Sherratt, N. A., Culvenor, J., Grubman, A., Furness, J. B., White, A. R., Finkelstein, D. I., Barnham, K. J., & Lawson, V. A. (2016). Restoration of intestinal function in an MPTP model of Parkinson's Disease. Sci Rep, 6, 30269. https://doi.org/10.1038/srep30269
Ellett, L. J., Revill, Z. T., Koo, Y. Q., & Lawson, V. A. (2020). Strain variation in treatment and prevention of human prion diseases. Prog Mol Biol Transl Sci, 175, 121-145. https://doi.org/10.1016/bs.pmbts.2020.08.006
Klemm, H. M., Welton, J. M., Masters, C. L., Klug, G. M., Boyd, A., Hill, A. F., Collins, S. J., & Lawson, V. A. (2012). The prion protein preference of sporadic Creutzfeldt-Jakob disease subtypes. J Biol Chem, 287(43), 36465-36472. https://doi.org/10.1074/jbc.M112.368803
Lawson, V. A., Furness, J. B., Klemm, H. M., Pontell, L., Chan, E., Hill, A. F., & Chiocchetti, R. (2010). The brain to gut pathway: a possible route of prion transmission. Gut, 59(12), 1643-1651. https://doi.org/10.1136/gut.2010.222620
Lawson, V. A., Silburn, K. A., Gorry, P. R., Paukovic, G., Purcell, D. F., Greenway, A. L., & McPhee, D. A. (2004). Apoptosis induced in synchronized human immunodeficiency virus type 1-infected primary peripheral blood mononuclear cells is detected after the peak of CD4+ T-lymphocyte loss and is dependent on the tropism of the gp120 envelope glycoprotein. Virology, 327(1), 70-82. https://doi.org/10.1016/j.virol.2004.06.012
Lawson, V. A., Vella, L. J., Stewart, J. D., Sharples, R. A., Klemm, H., Machalek, D. M., Masters, C. L., Cappai, R., Collins, S. J., & Hill, A. F. (2008). Mouse-adapted sporadic human Creutzfeldt-Jakob disease prions propagate in cell culture. Int J Biochem Cell Biol, 40(12), 2793-2801. https://doi.org/10.1016/j.biocel.2008.05.024
Ugalde, C. L., Lewis, V., Stehmann, C., McLean, C. A., Lawson, V. A., Collins, S. J., & Hill, A. F. (2020). Markers of A1 astrocytes stratify to molecular sub-types in sporadic Creutzfeldt-Jakob disease brain. Brain Commun, 2(2), fcaa029. https://doi.org/10.1093/braincomms/fcaa029
Vella, L. J., Sharples, R. A., Lawson, V. A., Masters, C. L., Cappai, R., & Hill, A. F. (2007). Packaging of prions into exosomes is associated with a novel pathway of PrP processing. J Pathol, 211(5), 582-590. https://doi.org/10.1002/path.2145
Research Projects
- Student projects
- Project 1. Should we worry about prion associated heart disease?
- Project 2. COVID-19 associated brain injury and inflammation
Faculty Research Themes
Infection and Immunology, Neuroscience
School Research Themes
Biomedical Neuroscience, Molecular Mechanisms of Disease
Key Contact
For further information about this research, please contact Head of Laboratory Associate Professor Vicki Lawson
Department / Centre
Unit / Centre
Lawson laboratory: Transmissible neurodegeneration
MDHS Research library
Explore by researcher, school, project or topic.