From viruses to venom: what is exochemistry?
"Exochemistry" is chemistry "on the outside"....outside the organism that produces it, that is. Venom is a classic example, because toxins are designed to work in the bodies of other organisms. Viruses too use exochemistry to achieve their infectious aims. Read today's post to see how an appreciation of exochemistry can help us understand the world around us.
From viruses to venom: what is “exochemistry”?
Welcome back for what we hope is another thought-provoking AVRU blog! We hope you’ve been staying safe and managing to be productive during the disruptions caused by the ongoing pandemic. This week, I’m going to introduce you to another general concept that will provide you with yet more evidence (as if you need it!) of how fascinating nature is. The concept is “exochemistry”, which might be a mouthful, but really just means “chemistry on the outside”. On the outside of what? The organism that produced it.

This rough-scaled snake (Tropidechis carinatus) is doing its best to inject some exochemistry into a red-eyed tree frog (Litoria chloris). Frogs can be difficult prey to swallow due to their powerful legs and venom can mean the difference between a full or empty stomach for the snake. Interestingly, whilst a rough-scaled snake can easily kill a dog (or even a human) with its bite, frogs seem remarkably resistant and struggles like this can go on for tens of minutes and are not guaranteed to end with a meal for the snake. Photograph by Stewart Macdonald.
It has been said that “life is chemistry”. Whilst we might all quite reasonably argue that life is a lot more than just chemistry (after all there’s physics, economics, history and PlayStation 4s), it’s certainly true that organisms are incredibly complex sets of chemical reactions. That includes you and me….unless you’re an AI, in which case move along Robocop, there’s nothing to see here! All those chemical reactions are the stuff of life. They’re what keeps us going, enabling us to take in materials and energy from the environment and turn it into more of ourselves, or use it for harvesting yet more materials and energy.
We’ve mentioned homeostasis before – it’s that set of regulatory processes (composed of chemical interactions and reactions) that literally keeps us being us. One of the things about relying on all this complex molecular activity to stay stable is that we need to protect it from the vagaries of the environment. For example, here in Melbourne the weather is all over the place – one minute it’s warm and sunny, the next minute it’s cold and wet – but these external changes in temperature do not result in corresponding changes within us. We mammals (again, I’m assuming no reptiloids are reading, but if they are this doesn’t apply to you) are “endotherms” – endo means “from inside” and therm means “temperature” – we make our own warmth (again, utilising chemical reactions). So, even when the air temperature in Melbourne drops 10 degrees in 10 minutes, our core body temperature stays pretty much the same (although we may need to eat a few more cookies to help our chemical furnace work overtime!). For this to be possible, we need to shield our internal chemistry from the environment – we need a wall between us and what’s outside that wall.
The first walls of this kind were the cell membranes of ancient single-celled organisms (like bacteria). We use the terms “cell membrane” and “cell wall” to refer to different structures, but for the moment this doesn’t matter - both are barriers between the chemical systems of a cell and the environment. As soon as the first cell membranes evolved to protect the vital chemical processes of the first organisms, the world was separated into inside and outside. This is the birth of organisms, which are (amongst other things) systems separate from, but interdependent with, their environments. Most of an organism’s chemistry takes place on the inside – whether it’s within the membrane of a single-celled organism or between the cells of a multicellular organism which are themselves encased in walls of tissue (like vessels and skin). This chemistry is an organism’s endochemistry. Organisms also produce molecules (chemicals), however, that are designed to function outside those walls – these are the exochemicals – exo means “on the outside”.
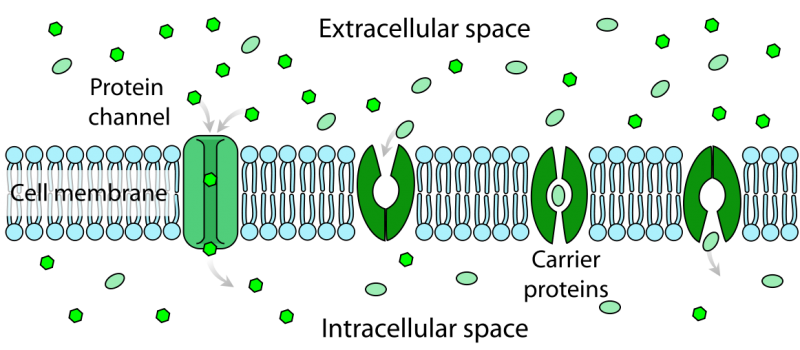 A schematic depiction of a cell membrane showing the intracellular space inside the membrane (site of endochemistry), the extracellular space outside the membrane (site of exochemistry for single-celled organisms) and the proteins channels and carriers that transport molecules from one side to the other. Wikimedia Commons.
A schematic depiction of a cell membrane showing the intracellular space inside the membrane (site of endochemistry), the extracellular space outside the membrane (site of exochemistry for single-celled organisms) and the proteins channels and carriers that transport molecules from one side to the other. Wikimedia Commons.
Single-celled organisms often have to secrete chemicals across their cell membranes to the outside in order to digest food, breaking it down into molecules that can then be transported back across the cell wall. Exochemistry was also central to the evolution of multicellular organisms – cells communicate with each other by sending chemicals across their membranes to receptors in the membranes of other cells. This sort of thing should be familiar to you if you read last week's post (Toxins: a primer by Bianca op den Rouw), and you should be starting to see where venom and the toxins it contains fit in. Toxins are, definitionally, molecules that are designed to function outside the body of the organism that produces them….because poisoning yourself isn’t a great idea! Toxins are exochemicals. Pheromones are another good example – these are chemicals that multicellular organisms (again, including us) use to communicate with each other. Have you ever thought your partner, parent or child just smelled really good, even though they weren’t wearing any perfume? Could be pheromones. Then again, modern humans seem to have lost their taste for some of the stinkier pheromones out there, which is why deodorants and perfumes are such big business.
OK, the title mentions viruses, and you’re probably wondering by now whether that was just click bait or if I am actually going to talk about something relevant to our current situation. Although I’d love to continue to keep you in suspense, the answer is yes, I am………now. Viruses are strange things that can make us question a lot of the categories we use to understand the biological world. Notice I referred to them as “things” and not as “creatures” or “organisms”. In fact, it has long been debated whether or not viruses should even count as being “alive”. A useful way to think about this is in terms of viruses being a “borderline case” – viruses are just the simplest systems that can do some of the things that organisms do, so simple that they must actually exploit the more complex systems of organisms to do those things. Whether or not viruses are “alive”, they are part of life because they are part of a functional continuum with living systems. The fundamental thing that organisms do is reproduce, and in order to reproduce a virus must hijack the reproductive (or rather “replicatory”, but let’s not worry about that now) machinery of an organism like us.
As you might imagine (or know first hand), we organisms are quite jealous of our reproductive machinery, and don’t exactly welcome our undead (“unalive”?) invaders. This is where viral exochemistry comes in. Our immune systems are on the lookout for marauders, so viruses have to use a chemical trick to avoid detection. A major way that our immune system recognises and neutralises invaders is by detecting particular sections of foreign proteins called “epitopes”. Viruses are sneaky – they evade the immune system by shielding the epitopes on their spike proteins with sugary molecules called “glycans”. In other words, they cover their outsides with chemical camouflage. The spike proteins themselves are also a classic exochemical – they are proteins that stick outside the protective envelope of the virus and interact with receptors on the surface of their host’s cells, fooling the cells into letting them in. Once past the protective outer membrane, the virus can start using the host cell’s copying processes to make more viruses. When enough viruses are made, the cell dies, releasing the viruses to go and infect more cells.
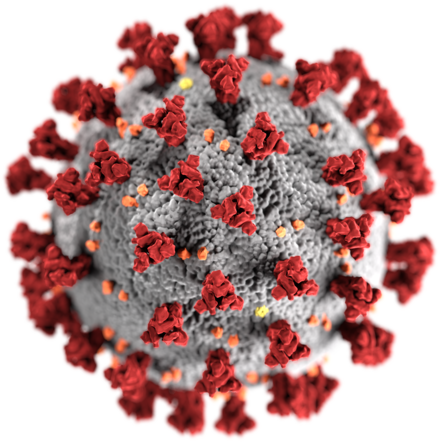 A computer-generated image of a coronavirus, with exochemical spike proteins coloured red. These proteins are responsible for the epithet - "corona" - because they give viruses imaged using an electron microscope an appearance similar to the corona of light surrounding the moon. Wikimedia Commons.
A computer-generated image of a coronavirus, with exochemical spike proteins coloured red. These proteins are responsible for the epithet - "corona" - because they give viruses imaged using an electron microscope an appearance similar to the corona of light surrounding the moon. Wikimedia Commons.
So, a viral spike protein binds a receptor in the host and subverts the host’s normal regulatory mechanisms in order to invade its cells and make more of itself. This is not so different from the sorts of things that the toxins which make up a venom do. A toxin may imitate an endogenous regulatory molecule, binding a receptor in the target organism and then subverting its normal function in some way. Toxins in venoms are designed to help the venomous organisms that produce them get a meal or deter another organism, but the ultimate goal for the venomous organism is the same as for any other living system – to be able to survive long enough and be healthy enough to make more of itself. Viruses cut straight to the chase – their spike proteins, chemicals they project to the outside of their protective envelopes, gain them access to cells, which allows them to make more of themselves. At the end of the day, making more of yourself (reproducing) is what life is about, in strictly reductive biological terms (I think we can all agree that life is about much more than this – getting likes on Instagram is important too!). Both venomous organisms and viruses use chemicals that they send outside themselves to achieve this basic aim. Both are textbook examples of exochemistry.
Stay tuned for more AVRU blog posts – we’ll be talking much more about toxins, exochemistry, venomous organisms and anything else even remotely related to venom. Thanks for reading and stay safe from those nasty viruses trying to hijack your baby-making machinery!
- Dr Timothy Jackson