Andrew Cox laboratory
-
Head of Laboratory

Dr Andrew Cox+61 3 8559 5439
Research Overview
In the Cox laboratory, we use zebrafish (Danio rerio) as a model system to study the metabolic regulation of growth during embryonic development, regeneration and cancer.
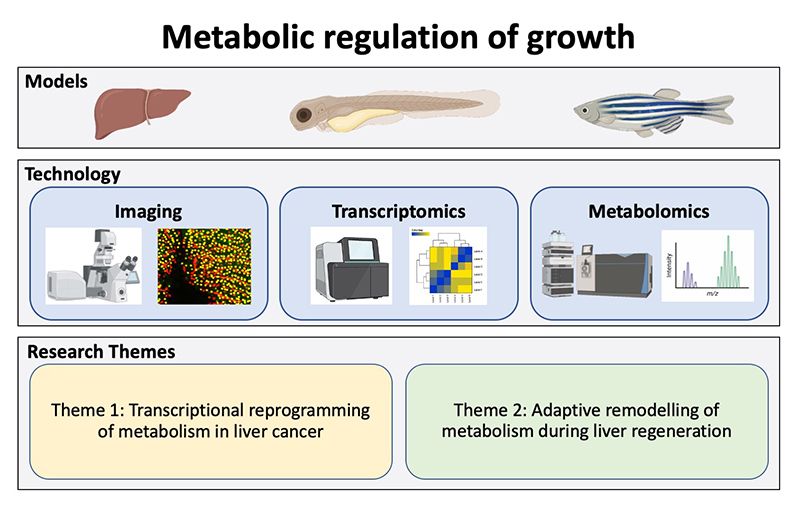
Hepatocellular carcinoma (HCC) is the most common form of liver cancer and among the most fatal cancer types. Although the pathophysiology of HCC has not been fully elucidated, the process clearly arises in the context of chronic liver disease brought on by environmental factors, which conspire with oncogenic pathways to initiate tumourigenesis. One of the emerging hallmarks of cancer is reprogramming of metabolism. Our lab takes advantage of cutting-edge technologies including multiphoton microscopy, metabolomics, transcriptomics and chemical genetic screens to elucidate the molecular underpinnings of metabolic reprogramming in cancer.
Staff
Anthony Karamalakis, Research Assistant
Athena Ong, Postdoctoral Fellow
Cerys Bladen, PhD Student
Madeline Webb, PhD Student
Mikaela Wong, PhD Student
Tara Tigani, PhD Student
Arina Abramovich, Hons Student
Collaborators
A/Prof Kristin Brown, Peter MacCallum Cancer Centre
Prof Ben Hogan, Peter MacCallum Cancer Centre
Prof Kieran Harvey, Peter MacCallum Cancer Centre
Prof Mark Dawson, Peter MacCallum Cancer Centre
Prof Malcolm McConville, University of Melbourne
A/Prof Louise Cheng, Peter MacCallum Cancer Centre
A/Prof Jan Kaslin, Monash University
A/Prof Kimberley Evason, University of Utah
A/Prof Matthew Steinhauser, University of Pittsburgh
Prof Wolfram Goessling, MGH – Harvard Medical School
Funding
View Dr Cox’s recent awards here (https://findanexpert.unimelb.edu.au/profile/792359-andrew-cox).
Research Opportunities
This research project is available to PhD students, Honours students, Master of Biomedical Science to join as part of their thesis.
Please contact the Research Group Leader to discuss your options.
Research Publications
Click here for the results of a PubMed search of Andrew's publications.
- Tan VWT, Salmi TM, Karamalakis AP, Gillespie A, Ong AJS, Balic JJ, Chan YC, Bladen CE, Brown KK, Dawson MA*, Cox AG*. SLAM-ITseq identifies that Nrf2 induces liver regeneration through the pentose phosphate pathway. Dev Cell. 2024 in press. DOI: 10.1016/j.devcel.2024.01.024. * Co-corresponding authors.
- Ong AJ, Bladen CE, Tigani TA, Karamalakis AP, Evason KJ, Brown KK*, Cox AG*. The KEAP1-NRF2 pathway regulates TFEB/TFE3-dependent lysosomal biogenesis. PNAS. 2023. May 30;120(22):e2217425120. DOI: 10.1073/pnas.2217425120. * Co-corresponding authors.
- Vaidyanathan S#, Salmi TM#, Sathiqu RM, McConville MJ, Cox AG*, Brown KK*. YAP regulates an SGK1/mTORC1/SREBP-dependent lipogenic program to support proliferation and tissue growth. Dev Cell. 2022 Mar 28;57(6):719-731.e8. DOI: 10.1016/j.devcel.2022.02.004. #Co-first author. *Co-corresponding author.
- Salmi TM, Tan V, Cox AG*. Dissecting metabolism using zebrafish models of disease. Biochem Soc Trans. 2019 Feb 28;47(1):305-315. DOI: 10.1042/BST20180335. * Corresponding author.
- Cox AG*, Hwang KL, Tsomides A, Miesfeld JB, Galli GG, O'Connor K, Yimlamai D, Chhangawala S, Yuan M, Lien E, Nissim S, Camargo FD, Asara J, Houvras Y, Link BA, Goessling W. Yap regulates glucose utilization and sustains nucleotide synthesis to enable organ growth. EMBO Journal. 2018. Nov 15;37(22). e100294. DOI: 10.15252/embj.2018100294. *Co-corresponding author
- Cox AG, Tsomides A, Kim AJ, Saunders D, Hwang KL, Evason KJ, Brown KK, Lien E, Chhangawala S, Lee BC, Nissim S, Dickinson BC, Chang CJ, Gladyshev VN, Asara J, Houvras Y and Goessling W. Selenoprotein H is an essential regulator of redox homeostasis that cooperates with p53 in development and tumorigenesis. PNAS. 2016. Sep 20;113(38):E5562-71. DOI: 10.1073/pnas.1600204113.
- Cox AG, Hwang KL, Brown KK, Evason KJ, Beltz S, Tsomides A, O'Connor K, Galli GG, Yimlamai D, Chhangawala S, Yuan M, Lien EC, Wucherpfennig J, Nissim S, Minami A, Cohen DE, Camargo FD, Asara JM, Houvras Y, Stainier DY, Goessling W (2016). Yap reprograms glutamine metabolism to increase nucleotide biosynthesis and enable liver growth. Nat Cell Biol. 18(8):886-96. DOI: 10.1038/ncb3389.
- Cox AG, Goessling W. The lure of zebrafish in liver research: regulation of hepatic growth in development and regeneration. Curr Opin Genet Dev. 2015 Apr 6;32:153-161. DOI: 10.1016/j.gde.2015.03.002.
- Cox AG, Saunders DC, Kelsey PB Jr, Conway AA, Tesmenitsky Y, Marchini JF, Brown KK, Stamler JS, Colagiovanni DB, Rosenthal GJ, Croce KJ, North TE, Goessling W. S-nitrosothiol signaling regulates liver development and improves outcome following toxic liver injury. Cell Reports. 2014 Jan 16;6(1):56-69. DOI: 10.1016/j.celrep.2013.12.007.
- Rosenbluh J, Nijhawan D, Cox AG, Li X, Neal JT, Schafer EJ, Zack TI, Wang X, Tsherniak A, Schinzel AC, Shao DD, Schumacher SE, Weir BA, Vazquez F, Cowley GS, Root DE, Mesirov JP, Beroukhim R, Kuo CJ, Goessling W, Hahn WC. β-Catenin-driven cancers require a YAP1 transcriptional complex for survival and tumorigenesis. Cell. 2012 Dec 21;151(7):1457-73. DOI: 10.1016/j.cell.2012.11.026.
Research Projects
- Transcriptional reprogramming of metabolism in liver cancer
- Adaptive remodelling of metabolism during liver regeneration
Faculty Research Themes
School Research Themes
Cancer in Biomedicine, Molecular Mechanisms of Disease
Key Contact
For further information about this research, please contact Head of Laboratory Dr Andrew Cox
Department / Centre
Unit / Centre
MDHS Research library
Explore by researcher, school, project or topic.