Melbourne Histology Platform

The Melbourne Histology Platform assists researchers to prepare animal, human, and plant tissue samples to investigate microanatomy using optical microscopy for analysis of basic biological processes and disease processes. We provide:
- Biological tissue preparation, processing, embedding, and sectioning
- Staining of biological tissues (including special stains and immunostaining)
- A consulting service for experimental design
- Access to sample sectioning and staining facilities for self-use
- Training in histological techniques
News
Upcoming events
Past events
-
Biological sample preparation winter workshops
 Event
Event -
Tuesday 10am - 12pmIntro To series 2023
 Event
Event -
Friday 12pm - 1pm2022 sample preparation "lunch and learn"
 Event
Event -
Thursday 12pm - 1pm2022 sample preparation "lunch and learn"
 Event
Event -
Wednesday 12pm - 1pm2022 sample preparation "lunch and learn"
 Event
Event -
Tuesday 12pm - 1pm2022 sample preparation "lunch and learn"
 Event
Event -
Monday 12pm - 1pm2022 sample preparation "lunch and learn"
 Event
Event -
MHP winter webinar series 2021
 Event
Event
With over 30 years' staff experience in routine and specialised histology techniques, the Melbourne Histology Platform is well equipped to handle uncommon samples with unconventional or challenging sectioning or staining needs.
As well as offering a full professional histology service, the Melbourne Histology Platform can provide full training in the required histological techniques, which researchers can then perform using the equipment and facilities available on a pay-for-use basis.

Services
We perform a range of core and advanced histology procedures including:
- Paraffin and frozen tissue embedding
- Paraffin, cryo-and vibro-microtome tissue sectioning
- Automated haematoxylin and eosin staining
- Routine and special staining including silver stains
- Antibody labelling/immunohistochemistry
- Sectioning and staining of a range of tissues including human, rodent, insect, plant and sea anenomes
Our technical staff have over 30 years’ combined experience in routine and specialised histology techniques.

Equipment
- Sakura Tissue Tek paraffin processors (staff operated)
- Sakura Tissue Tek paraffin embedders
- Microm paraffin microtomes
- Leica CM1900 cryostat
- Leica CM1860 cryostats
- Leica VT1000 S vibrating blade microtome
- Leica autostainer XL and coverslipper (staff operated)
- Dako Link 48 automated immunostainer (staff operated)
- Milestone BoneSTATION sample decalcification system
- Primera slide printer
- Pulsar cassette printer
We work in collaboration with key University microscopy services including the Biological Optical Microscopy Platform and the BioSciences Microscopy Unit, and in collaboration with the Phenomics Australia Histopathology and Digital Slide Service.

Complementary Platform Technologies
For more information about the wider platform community, please visit Research Website, which is available to all users.
Details about the University’s research infrastructure related resources and services can be found on Research Gateway, which is available to the University of Melbourne staff and Graduate Researchers.
_
Staff

Dr Chris Freelance
Platform Manager
Dr Chris Freelance is an evolutionary biologist whose research spans comparative morphology, sensory ecology, and neuroscience, and has involved the utilisation, development and refinement of techniques in invertebrate histology and microscopy. As the Platform Manager, Chris provides consultations, facilitates engagement and researcher education events, develops MHP's strategy, and oversees the Platform's staffing and physical infrastructure.

Laura Leone
Laboratory Coordinator
Laura has extensive experience in diagnostic and research histology. She has worked at The University of Melbourne for over a decade where she has encountered a range of samples ranging from brains and livers to leaves, sea anemones and grasshopper eggs!

Lisa Foster
Histology Technical Officer
Lisa completed a Bachelor of Arts/Science with Honours in the Department of Anatomy and Neuroscience at The University of Melbourne. With a research background, she enjoys assisting clients with their histological techniques and integrating her knowledge and expertise into a variety of projects.

Connor Daymond
Histology Technical Officer
Connor is a PhD Candidate whose research entails neurology, virology, and biomarker identification. His research involves the utilisation of histological techniques to assess pathological changes in brain tissue. As a Histology Technical Officer, he uses his research experience to assist clients and platform staff with histological projects.

Trixie Pineda-Dator
Histology Technical Officer
Trixie, who has a Bachelor’s degree in Medical Laboratory Science has had vast experience in diagnostic histology, special staining, and immunohistochemistry. She uses her knowledge and expertise to assist clients with their histological projects and techniques.
_
Governance
Platform Advisory Committee
Members
Dr Will de Nardo – Chair; Dept of Anatomy and Physiology
Prof Andrew Pask – Deputy Chair; School of BioSciences
Dr Chris Freelance – Platform Manager, Melbourne Histology Platform
Prof Erica Fletcher – Academic Lead, Melbourne Histology Platform; Dept of Anatomy & Physiology
Dr Richard Frampton – School of Biomedical Sciences
Prof Stephen Rogerson – Melbourne Medical School and Peter Doherty Institute
Ms Tina Cardamone – Phenomics Australia Histopathology and Slide Scanning Service
Ms Rita Paolini – Melbourne Dental School
Secretariat
Ms Vanessa Felice – School of Biomedical Sciences
_
Contact us
- Address
- E214 – 216, Level 2 East, Medical Building (181)
The University of Melbourne
Parkville VIC 3010 - biomedsci-histology@unimelb.edu.au
- Phone
- +61 3 8344 5752

The [Platform] undertook some strange requests from me to cut and stain slides for an artistic project, a fringe festival show performed live on a microscope – they immediately worked out what I needed and spent the time with me to embed and cut the slides the specific way they needed to look, and was able to come up with unconventional solutions to help me achieve the right colours in the tissues. We now have a showcase of slides ready to share the beauty of the microscopic world with the public!
Clare Hampson
Creator/Performer, Alice Looking Through the Glass, Melbourne Fringe 2017
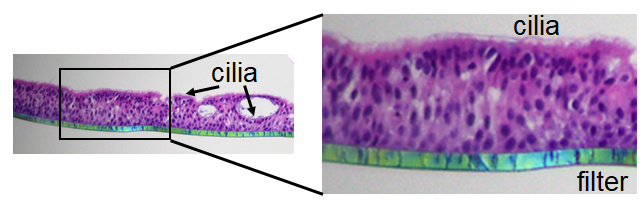
Our research involves working with primary human nasal epithelial (HNE) organoids. It was a challenge to preserve the complex tissue architecture of air-liquid interface (ALI) differentiated HNE. The [Platform] helped us work out a immunohistochemistry protocol and embed and cut the slides – the H&E staining revealed the complexity of the pseudostratified epithelium with apical cilia and the presence of spheroids embedded within the epithelium with luminal cilia. The ALI-HNE recapitulate key features of the nasal epithelium, making them the perfect model for human respiratory infections, including SARS-CoV-2, the cause of COVID-19!
Elizabeth Vincan (Professor) and Bang Tran (Research Fellow)
Department of Infectious Diseases, Melbourne Medical School, University of Melbourne
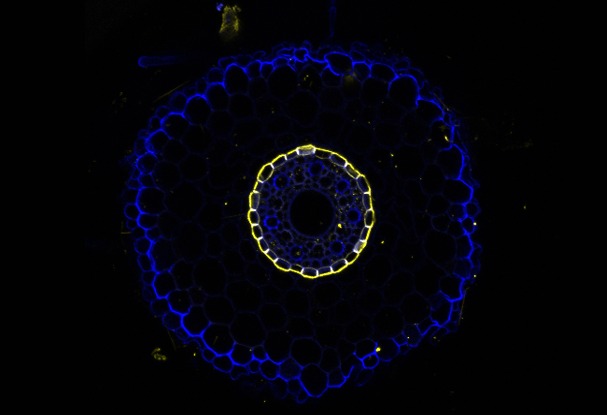
The root endodermis serves as a check-point for the passage of nutrients and toxicants from the soil into the vascular system of the plant. There is little known about how the secondary cell walls composed of suberin (yellow) and callose (blue) respond to nutrient deficiency and salinity.
The Melbourne Histology Platform assisted me to develop the method to section barley and rice roots using a vibratome and to develop a staining protocol. Using these images, we will discover how (i) root barrier development is regulated by nutrient supply and sodium toxicity and (ii) how root barriers mediate nutrient uptake and sodium exclusion.
Vanessa Melino
Research Fellow, School of Agriculture and Food, University of Melbourne
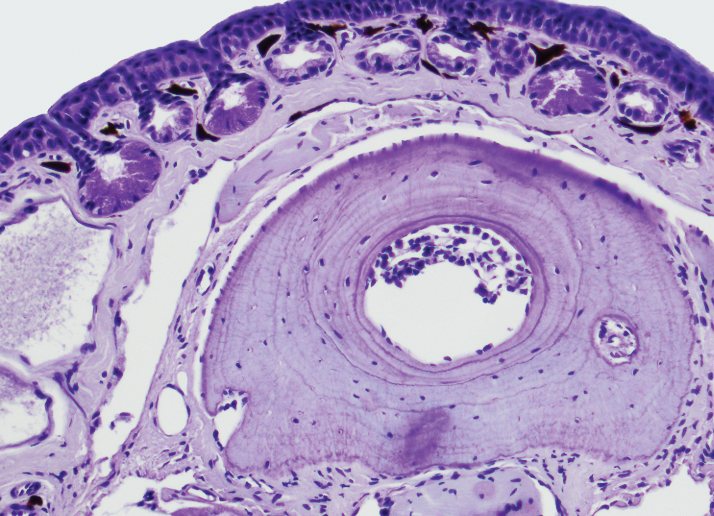
My research investigates the relationship between age and survival of populations of the critically endangered spotted tree frog Litoria spenceri impacted by chytrid fungus. We collect toe clips to use growth lines in the toe bones to age the frogs, tracking how the age structure of populations is changing with threats over time.
The Platform assisted me with processing and embedding my samples and provided training for using their paraffin microtomes to section the tissue. The staff provided guidance for adjusting my sectioning technique and for refining the staining to identify growth ring formation in periosteal bone.
Sophie McCulloch
Master of Science student, School of BioSciences, University of Melbourne
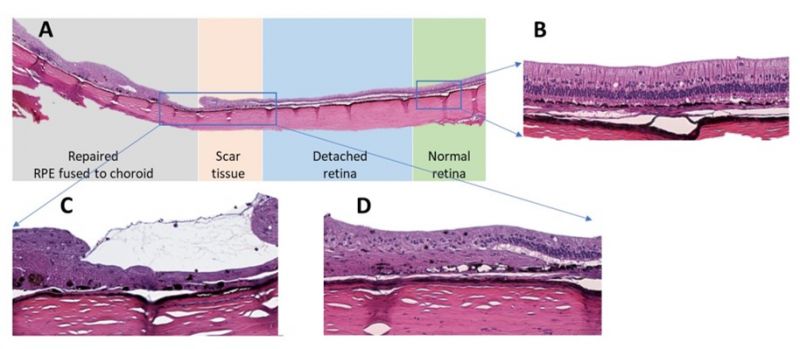
This project involved a collaboration between the Department of Optometry and Vision Sciences, (The University of Melbourne) and the Centre for Eye Research Australia (CERA) to develop a new surgical device called Retinal Thermofusion (RTF) that was validated in a rabbit retinal detachment model. Rabbit eyecups were processed by the Melbourne Histology Platform to give retina cross-sections stained with H&E. As shown above, this allowed us to distinguish between regions of normal retina (B), transition between repaired and detached retina (D) and within repaired retinal hole (C). The cross-sections provided us with histopathological evidence of successful retinal reattachment post surgery as indicated by fusion of the RPE to the choroid tissue (C).
Anh Hoang (Research Assistant) and Bang Bui (Associate Professor)
Department of Optometry and Vision Sciences, University of Melbourne
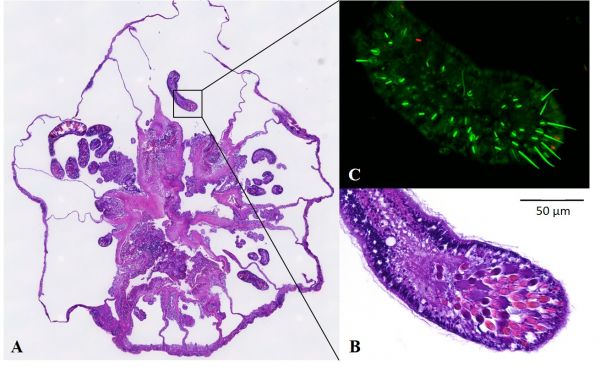
At the Marine Microbial Symbiont Facility, we study the microbial communities of the anemone E. pallida. We use fluorescence in situ hybridization (fISH) to localise bacteria associated with anemones and their microalgal endosymbionts. The Melbourne Histology Platform processes and sections our anemones into 80+ transverse sections and provides haematoxylin & eosin stained sections flanking unstained sections for fISH. These sections have allowed us to identify microbial aggregates associated with the anemones’ acontia: a defensive tissue rich in stinging cells (A-C).
Ashley Dungan
PhD candidate, School of BioSciences, University of Melbourne
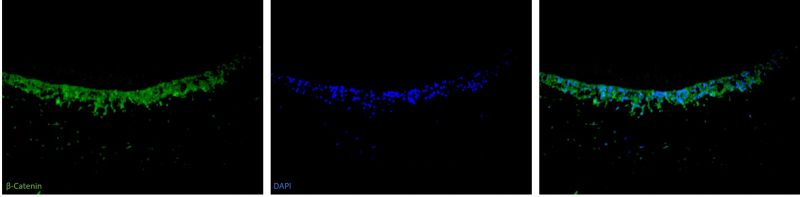
My research investigated the dental pulp and odontoblast cell’s response to dental caries in normal and hypomineralised teeth. The Melbourne Histology Platform assisted with the immunofluorescent (IF) staining of sections of my teeth samples with the dentine-formation biomarkers β-Catenin and Dentine Matrix Protein 1. The Platform staff provided training and helped with the optimisation to develop a IF protocol for my biomarkers. Their equipment and expertise helped me achieve the optimal staining needed for my project.
Janita Shah
Doctor of Clinical Dentistry (Paediatric Dentistry) student, Melbourne Dental School, University of Melbourne
_
The iLab booking system
From October 2020, the Melbourne Histology Platform is using an iLab booking system. This system manages all equipment bookings for trained users, as well as requests for quotes, consultation, training and service provision.
Important points
- Supervisors/lab heads must register for an iLab account prior to their researchers/lab members registering for iLab. Registration instructions can be found in the Quick Reference Guides below.
- From Monday 7th December 2020, it will be mandatory to provide financial information for billing when submitting service requests or making an instrument reservation. This information is a valid Themis string for University of Melbourne users, or a purchase order number for external users.
- On Saturday 1st May 2021, iLab financial integration with Themis was completed. Principal investigators are no longer able to manually add a Themis string to iLab. If a UoM PI wants to to pay for services booked through iLab using a Themis string that is not available to them in iLab, they can submit a ServiceNow request to have the string made available.
Quick reference guides
The Quick Reference Guides provide an overview of how to perform tasks in the Booking System, including registering for an account, submitting requests and booking resources.
For Principal Investigators/Laboratory heads: this guide contains information for laboratory heads who need to manage Platform usage by their lab members or manage financial information. This QRG also provides instructions on how to delegate the 'Lab Manager' responsibility to one of your lab members.
For Researchers: this guide contains information for researchers who use the platform.
FAQs: this guide answers frequently asked questions about using the iLab booking system.
Requesting Services: this guide helps researchers to select which form to use to request the services they are seeking.
Seeking support
If answers to your questions about using the MHP booking system cannot be found in the Quick Reference Guides, please contact MHP staff for assistance.
Melbourne Histology Platform terms of access
The Melbourne Histology Platform is open to researchers from The University of Melbourne as well as researchers from other Australian universities or research institutions. Use of facilities or equipment is on a pay-for-use basis and is subject to the researcher first completing the relevant inductions and training with the Platform staff.